
He found that the amount of adenine was very close to equaling the amount of thymine, and the amount of cytosine was very close to equaling the amount of guanine, or A = T and G = C. Around this time, Austrian biochemist Erwin Chargaff 5 (1905–2002) examined the content of DNA in different species and discovered that adenine, thymine, guanine, and cytosine were not found in equal quantities, and that it varied from species to species, but not between individuals of the same species. What is meant by the 5ʹ and 3ʹ ends of a nucleic acid strand?īy the early 1950s, considerable evidence had accumulated indicating that DNA was the genetic material of cells, and now the race was on to discover its three-dimensional structure.Pyrophosphate is subsequently hydrolyzed, releasing the energy used to drive nucleotide polymerization. The two unused phosphate groups from the nucleotide triphosphate are released as pyrophosphate during phosphodiester bond formation. The resulting strand of nucleic acid has a free phosphate group at the 5ʹ carbon end and a free hydroxyl group at the 3ʹ carbon end. 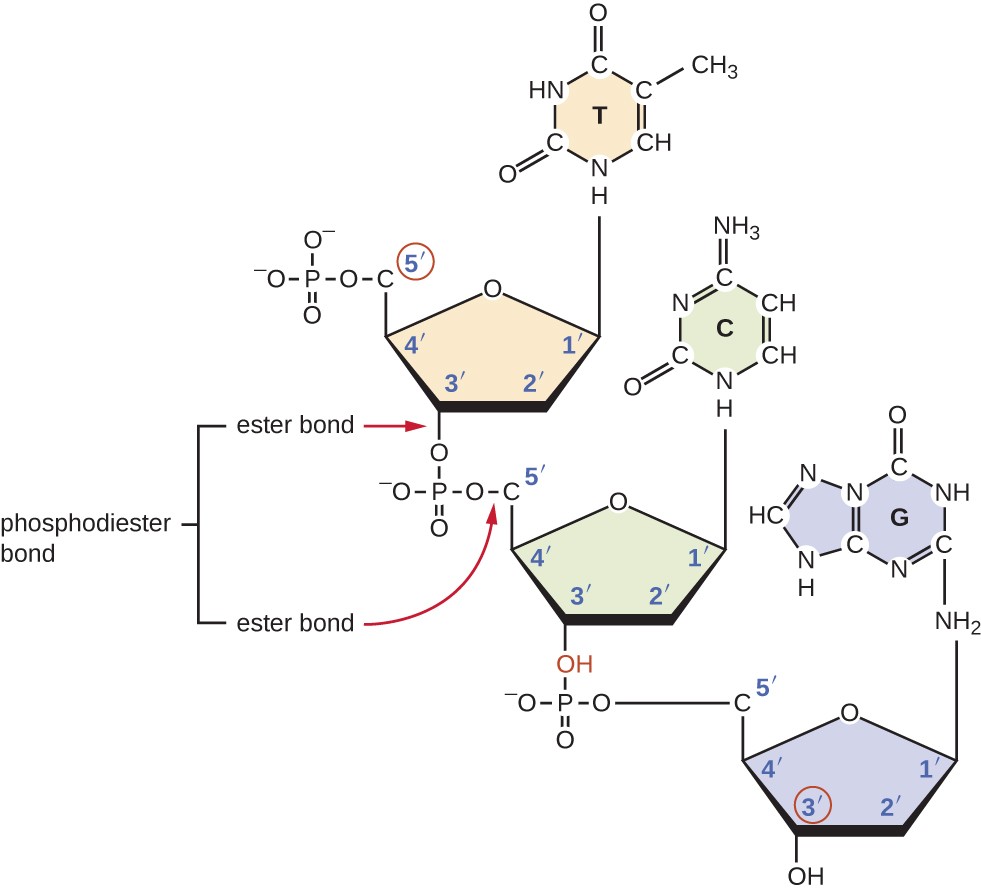
To construct the sugar-phosphate backbone, the two terminal phosphates are released from the dNTP as a pyrophosphate. During the polymerization process, deoxynucleotide triphosphates (dNTP) are used. Phosphodiester bonding between nucleotides forms the sugar-phosphate backbone, the alternating sugar-phosphate structure composing the framework of a nucleic acid strand ( Figure 10.13). Individual nucleoside triphosphates combine with each other by covalent bonds known as 5ʹ-3ʹ phosphodiester bonds, or linkages whereby the phosphate group attached to the 5ʹ carbon of the sugar of one nucleotide bonds to the hydroxyl group of the 3ʹ carbon of the sugar of the next nucleotide. A nucleoside comprises the five-carbon sugar and nitrogenous base.įigure 10.12 Nitrogenous bases within DNA are categorized into the two-ringed purines adenine and guanine and the single-ringed pyrimidines cytosine and thymine. The carbon atoms of the five-carbon deoxyribose are numbered 1ʹ, 2ʹ, 3ʹ, 4ʹ, and 5ʹ (1ʹ is read as “one prime”). The three components of a deoxyribonucleotide are a five-carbon sugar called deoxyribose, a phosphate group, and a nitrogenous base, a nitrogen-containing ring structure that is responsible for complementary base pairing between nucleic acid strands ( Figure 10.11). Nucleotides that compose DNA are called deoxyribonucleotides. The building blocks of nucleic acids are nucleotides. In this section, we will discuss the basic structure and function of DNA. In Mechanisms of Microbial Genetics, we will discuss in detail the ways in which DNA uses its own base sequence to direct its own synthesis, as well as the synthesis of RNA and proteins, which, in turn, gives rise to products with diverse structure and function. The base sequence of deoxyribonucleic acid (DNA) is responsible for carrying and retaining the hereditary information in a cell. Each nucleic acid strand contains certain nucleotides that appear in a certain order within the strand, called its base sequence. Like other macromolecules, nucleic acids are composed of monomers, called nucleotides, which are polymerized to form large strands. In this chapter, we will discuss the genetic role of a fourth class of molecules: nucleic acids. In Microbial Metabolism, we discussed the microbial catabolism of three classes of macromolecules: proteins, lipids and carbohydrates. Explain why the double helix of DNA is described as antiparallel.Identify the base pairs used in the synthesis of deoxyribonucleotides.Describe the biochemical structure of deoxyribonucleotides.By the end of this section, you will be able to:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
